Automatic DICOM Deidentification
Removes both burnt-in and DICOM metadata PHI. Compliant with Part 15 of the DICOM standard, HIPAA, and SOC 2 Type 2 — built for secure research, AI training, and external data sharing.
What it does.
Every DICOM file contains personal health information (PHI) in two distinct locations: structured metadata tags (patient name, MRN, date of birth, referring physician, institution, and so on) and burnt-in annotations — pixel-level overlays written directly onto the image by the scanner's acquisition software. Removing only one layer is insufficient. InVision's Automatic DICOM Deidentification pipeline handles both.
The tool is built for secure research data sharing, external AI training datasets, and multi-site collaborations where regulatory compliance is non-negotiable. It is compliant with Part 15 of the DICOM Digital Imaging and Communications in Medicine standard, HIPAA, and SOC 2 Type 2 — the same compliance framework that underpins InVision's clinical AI products.
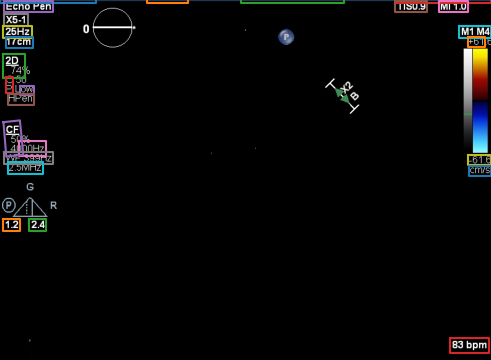
Deidentification runs as a fully automated step on studies leaving your clinical environment, with no manual review required. Clinical imaging data — chamber outlines, ultrasound cones, Doppler overlays — is preserved in full fidelity. Only the PHI is removed.
Four layers of protection.
Built for secure workflows.
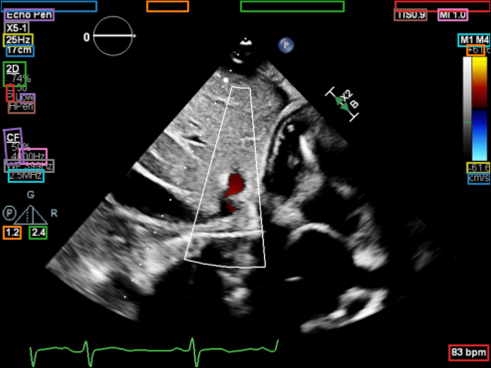
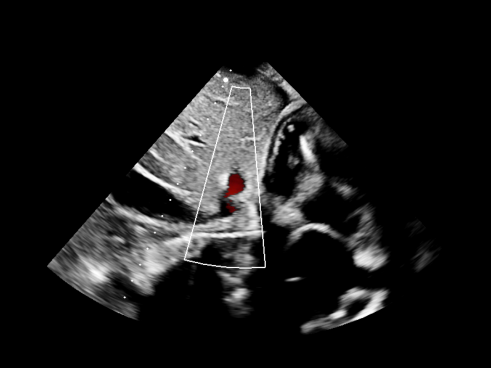
Example deidentified DICOMs.
Illustrative examples of echocardiographic studies processed through the automatic deidentification pipeline. These visuals represent how clinical imaging can be preserved while structured metadata and burnt-in PHI are removed.
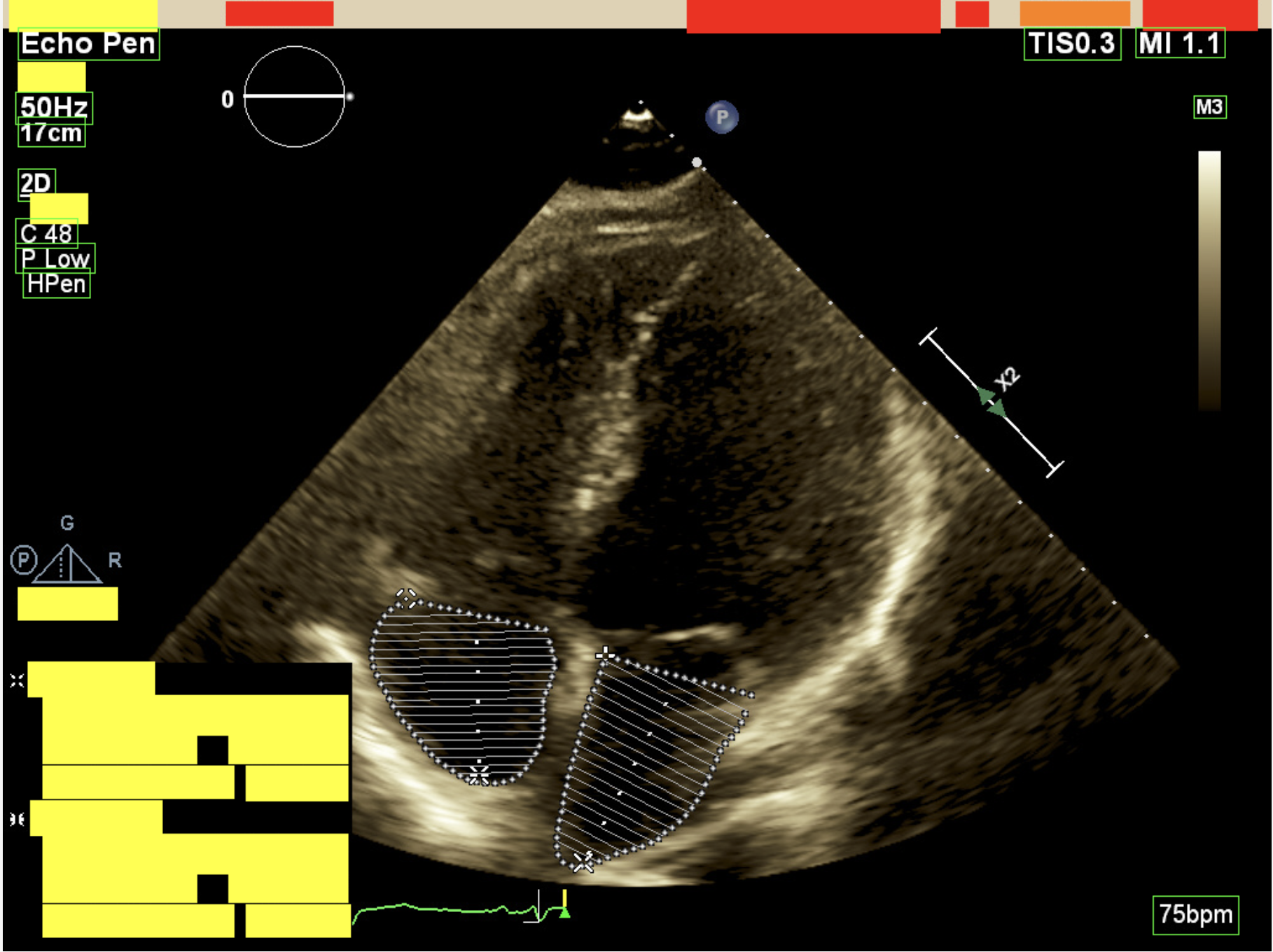
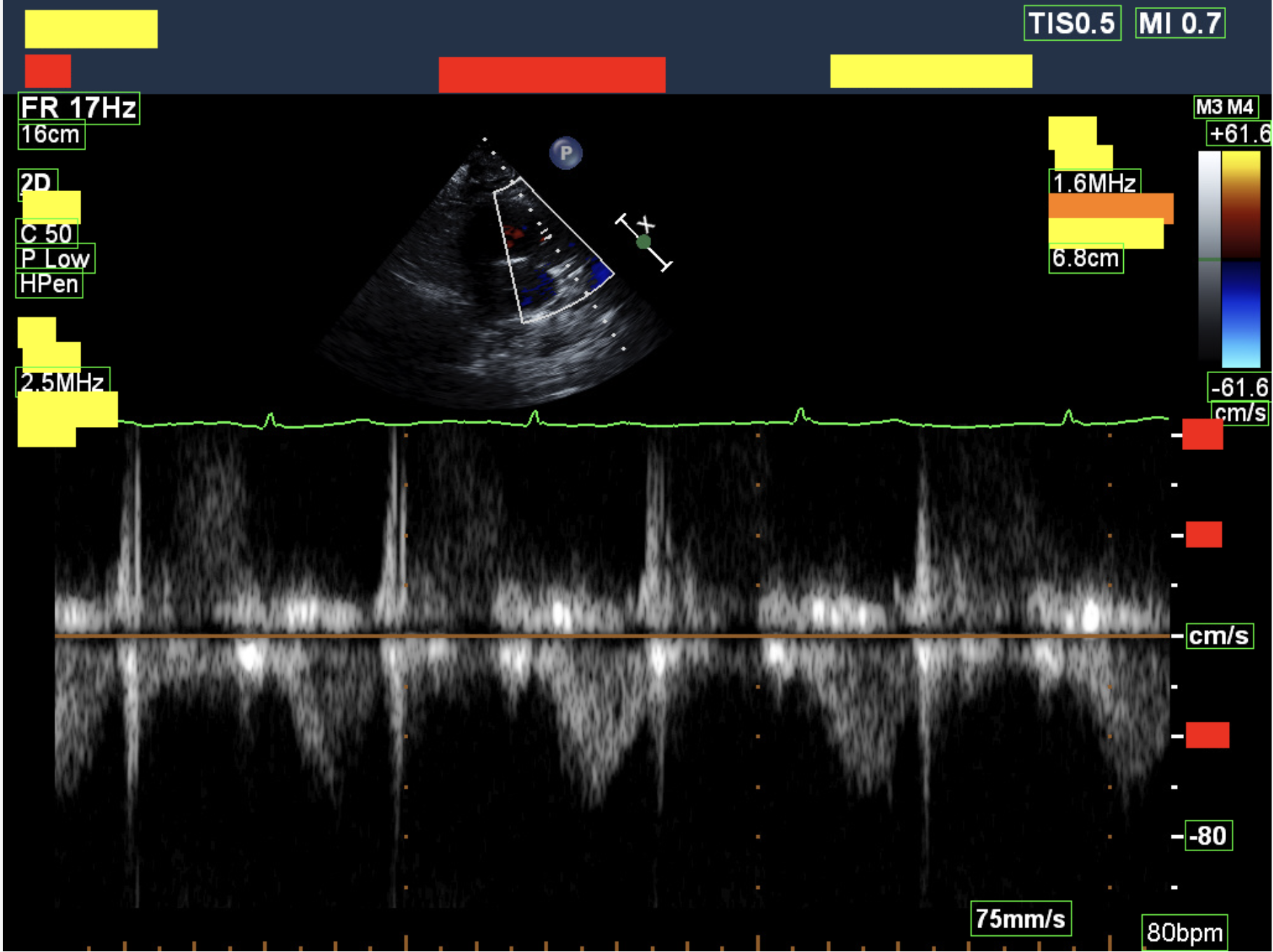
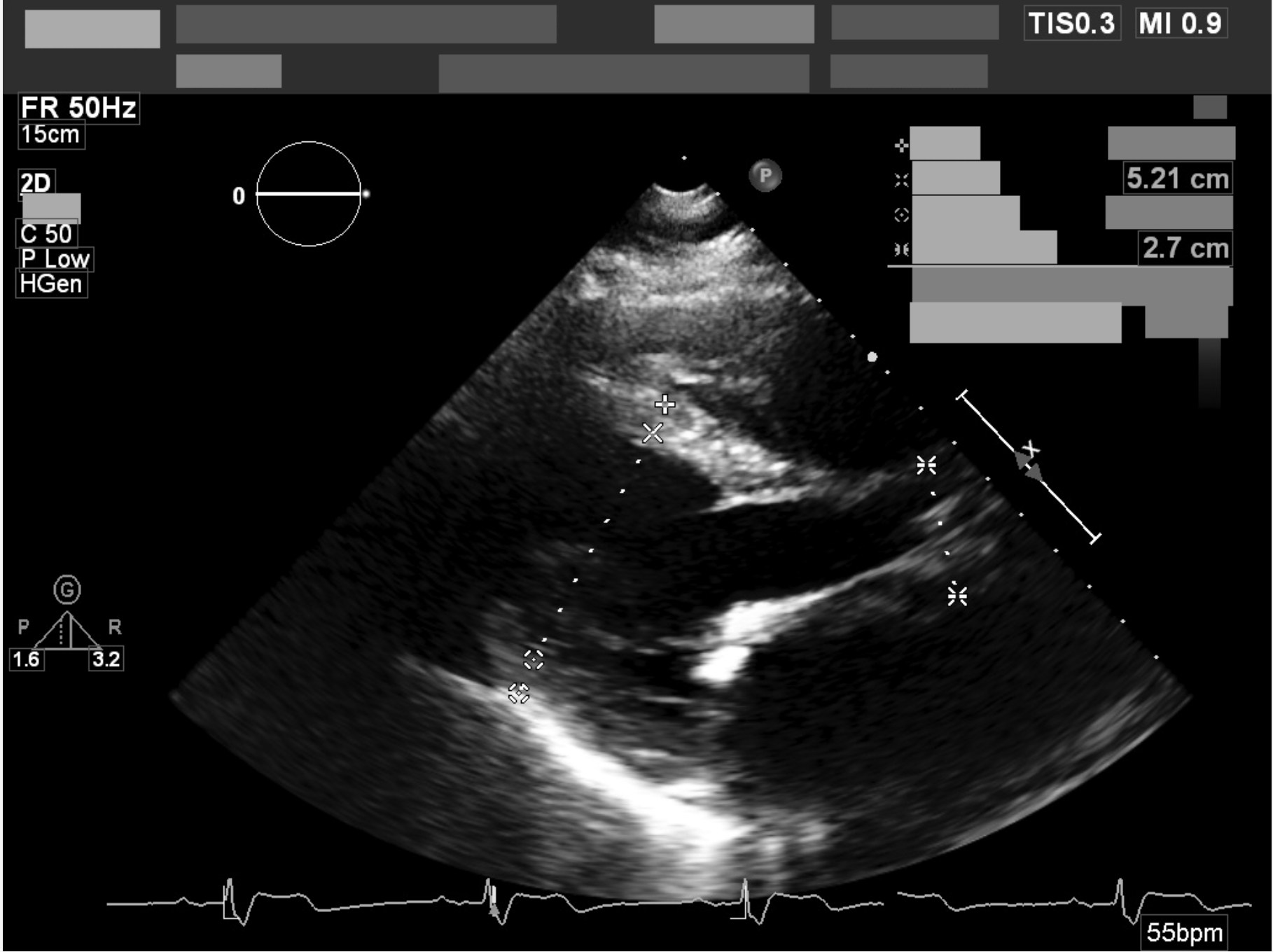

Removing annotations baked into pixels.
Burnt-in annotations — patient name, MRN, study date, and institutional headers written directly into the image by the scanner — cannot be removed by metadata scrubbing alone. These illustrative crops show representative pixel-level redaction while preserving surrounding clinical context.